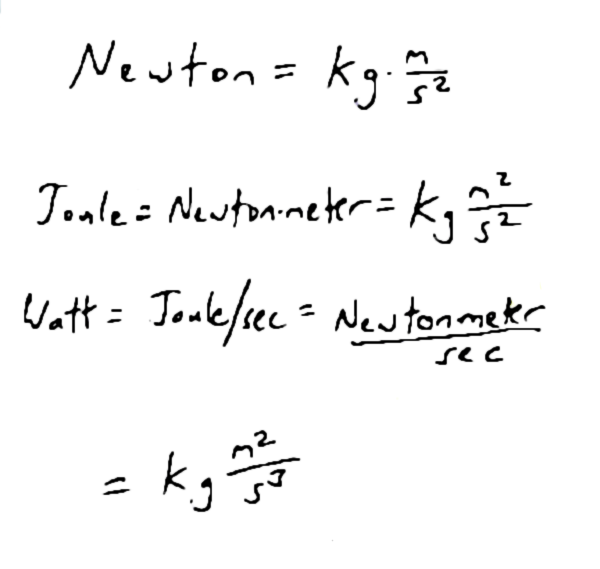Here we go again, again. It’s Monday‒the last one in April this year‒and I’m writing another effing blog post.
I keep trying weird little things in the hope that they engender or otherwise encourage something positive in my life. For instance, after briefly using a blue Bic® Round Stic™ pen on Friday, I realized that I had on some level missed writing with them.
I wrote Mark Red and The Chasm and the Collision, and the “short story” Paradox City all with blue and/or black medium Bic™ Round Stic® pens. These were the only ones available through commissary up at FSP. After a while, the guys who did tattoos would just give me new ones to use as long as I gave them back when empty/traded an empty one for the new one, so they could use them to make tattoo guns, and I went through such pens pretty quickly.
I thought to myself (since I have trouble thinking to anyone else*) that maybe if I started using these pens regularly again, I might help give myself the energy to start doing some new fiction writing. So, I ordered a box of them, which is at least quite inexpensive, and I have one in my pocket now.
It’s a fairly childish notion, perhaps, but just because something is childish does not mean it’s wrong or bad. Adults get rid of too many childish things‒sometimes on the advice of effing Saul of Tarsus of all the pathetic losers to whom to listen‒and adopt too many “adultish” things that are no more sensible, not as rewarding, and are reliably productive of negative outcomes.
Of course, some childish things do need to be left behind. Ideally, one does not want to keep believing in Santa Claus or monsters in the closet or that stepping on a crack will break your mother’s back any longer than one must. Wetting the bed is also worth stopping as early as one can.
But it can be good for one to keep asking questions about how things work and what they are and what they do and how they got to be the way they are, and being delighted in seeing and learning new things, and enjoying simple games and going outside and stuff like that.
Anyway, I doubt this particular choice of pens will actually get me to write any fiction again, but maybe it will at least feel good to use them again for a while.
As you know, I have at least a few stories, such as Outlaw’s Mind and The Dark Fairy and the Desperado that I have started that I’d like to finish, and I have some other stories on the back burner that I’d like to start and write. If I could just find a patron to support me while I write, so I didn’t have to do anything else, I could probably do it. But despite its name, even Patreon doesn’t really work that way.
People who support “creators” on Patreon pay regular, specified amounts and expect regular, piecemeal output (like daily blogs, for instance, though being the intellectually stunted populace that we are, people more often seem to want video stuff). If I put up a Patreon, or a “Go fund me” thing (whatever the proper term for that is) I doubt that I would get a lot of people supporting me and just waiting while I work on a long form writing project.
If anyone wants to do that, and is able to do it, let me know. Just remember, I’m slightly paranoid, so I will probably suspect some scam at first if you approach me‒unless I already know you, of course.
All of this is really just fantasizing, obviously. I might as well request that the person who wants to be my patron for writing fiction is also a beautiful woman who is just my type (whatever that might be) and who wants to be in a long-term relationship with me. Oh, and also, she owns a dragon, as well as an FTL spaceship. Hey, maybe she’s a Time Lord and has her own TARDIS!
Actually, if I had the use of a TARDIS, it would probably distract me completely from writing fiction. But I probably wouldn’t spend as much time (har) just traveling and having adventures as most of, for instance, the Doctor’s companions do. I would want to learn how this technology works!
I don’t understand why none of the people who enter the TARDIS and gape at the whole “bigger on the inside” thing don’t right then and there ask how it works! (Occasionally some do so, rather halfheartedly).
And when the trite little, dismissive answers such as Nardole gives are offered, they should say, “No, no, I mean how does it actually work? What is the science and technology involved, how is it carried out and maintained? What is the physics underlying it, how was it discovered, how was it harnessed? Do you have any primers on that, any online courses, any textbooks, even any ‘how does it work’ for kids books? And for that matter, how does the time stream and everything work, how is it traversed, what is the physics behind the functioning of the TARDIS? We’ll get to the biology of regeneration in due time, but I want to understand all this. To Hell with going and fighting Daleks or whatever, you can literally do that whenever you feel like, because you have a time machine!”
I guess it wouldn’t be a very fun show, just to watch someone studying Time Lord science and technology, but in real life, if I had access, I like to think that’s how I would spend a lot of my time. And I think I think correctly.
All right, that’s enough stupid fantasizing for today, wouldn’t you say? None of those or any other good things are likely to happen to me (some are far more probable than others, but none are worth betting on).
I am much more likely to keep developing new and harder to control pain and more frequently recurring and persistent pain and greater and greater frustration and despondency and depression until finally, at long last, it kills me. Then, at least, everyone in the universe overall will be just a little bit happier. On average, anyway.

*Though in a certain sense, this blog is an instance of me thinking to other people. But that requires the other people to be active participants, and it certainly cannot be done all day every day or any such thing.