I had a moment of idle curiosity this morning just before starting to write this. I recalled the bit of trivia that the average human power output/consumption is something around 80 or 100 Watts. I wasn’t sure which was more typical, but it doesn’t really matter; the numbers are well within the same order of magnitude, despite having nominally different numbers of digits.
Anyway, I decided to convert that into kilocalories* per day, just to confirm that the typically described numbers match up, because if they don’t, then something very strange is going on.
A Watt is a joule per second**, so to figure out how much energy output (in joules) there is in or from a human per day, you just multiply the watts times the number of seconds in a day (24 hours per day x 60 minutes per hour x 60 seconds per minute, or 86,400 seconds per day). Multiply that by the above-noted wattage and you get between about 6 and 8 million joules per day.
Now, there are 4,184 joules per kilocalorie, so dividing that into the number of joules yields: roughly between 1600 and 2000 kilocalories a day, which matches the data on basal metabolic rates. Neat.
Of course, they must match up, otherwise there would clearly be some major logical inconsistencies in our understanding of such thermodynamicalish matters. I don’t suspect that such a mismatch would have survived the scrutiny of scientists much longer than a snowball would last in a blast furnace; in other words, I consider textbook level physics to be pretty darn reliable. Nevertheless, it is good occasionally to check even such basic things, just to confirm for yourself that your understanding of reality is internally consistent and consistent with that which is measured and described by other people.
This is not to say that I worry about whether my “reality” is significantly different than that of other people. I don’t. While I have no doubt that the specific details of my personal experience are unique, this is so only in rather trivial ways.
I’ve not encountered any occurrence or argument that made me doubt whether everyone around me is subject to the same laws of physics as those to which I am subject. Of course, if tasked or merely bored, I can conceive of ways in which all that I think I know is illusory and/or delusional, as in the argument that precedes the cogito in Descartes’s most famous (non-mathematical) work.
With a bit of effort, one can almost always imagine ways in which the world could be deeply different than it seems. I’ve been known to do that at length‒indeed, at book length‒myself. But the fact that a thing can be imagined is not a reason, by itself, to promote a concept into “might actually be true” space. Presumably, there are limitless such things that could be imagined, but almost by definition (at least as I am using the word) there is only one reality.
Reality, as far as I can see, cannot contradict itself; actual paradoxes cannot be instantiated. I’d probably be prepared to bet my life on those propositions. But even if reality could contradict itself, that would also be a fact about reality. Whatever reality is, it is.
That’s trivial, of course, but sometimes it’s good to be reminded of the trivial things that one carries in one’s background knowledge but rarely considers or reconsiders‒things like the interchangeability of measures of energy and power and heat between different units.
With that full circle moment, I’m going to finish for today. I’m still very tired, and I’m rather discouraged and despondent and probably other d-words as well. This blog is all I really do, anymore, but my energy is lagging even for this. At least I don’t need to do payroll today, since I had to get it done early yesterday…which fact I found out yesterday.
Oh, well. Please do what you can to have a good day. And remember, there is no do or do not. There is only try.
*This is what we call “calories” when speaking of human energy intake and output, but a single “true” calorie is the amount of energy (heat) required to raise the temperature of 1 gram of water 1 degree centigrade (or, well, Kelvin if you want to be pedantish). A kilocalorie, or what we commonly call a calorie, is enough to raise a kilogram of water 1 degree Kelvin.
**A joule being the unit of energy in “SI” units. A joule (energy) is the integral of force with respect to distance, or a Newton-meter. A Newton is the measure of force, and is a kilgram-meter/ second-squared. So joules have the units kilogram-(meter squared)/second squared. Watts (a measure of power, or energy per unit time) are joules per second, which fact gives us the fun, lovely phenomenon of having cubic seconds in the denominator of the equation!
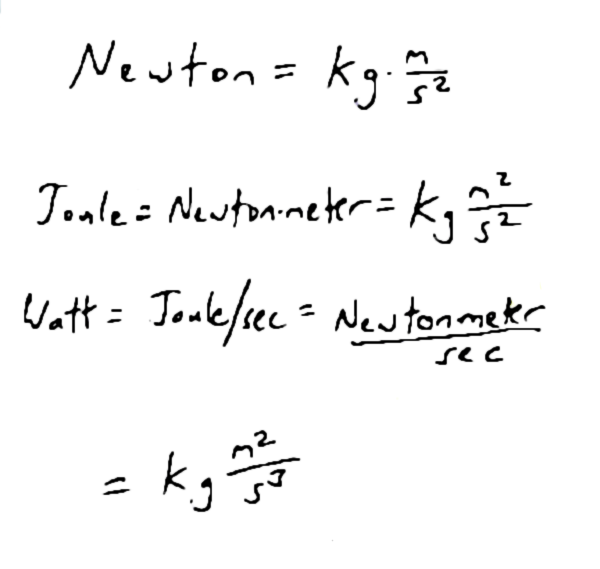


“much longer than a snowball would last in a blast furnace”
Unless it is hafnium carbide snow, which melts at 3928 degrees C, which could easily resist a blast furnace (about 1510 C). 🙂
Can you make “snow” out of Hafnium carbide? There must be SOME form of crystal that it forms that could be made into a kind of snow. But how would it ever get into the atmosphere to precipitate into snowflakes? Maybe somewhere deep down in Jupiter?