I woke up very early today‒way too early, really. At least I was able to go to bed relatively early last night, having taken half a Benadryl to make sure I fell asleep. But I’m writing this on my phone because I had to leave the office late yesterday, thanks to the hijinks of the usual individual who delays things on numerous occasions after everyone else has gone for the day. I was too tired and frustrated to deal with carrying my laptop around with me when I left the office, so I didn’t.
I’m not going to get into too much depth on the subject, but I found an interesting article or two yesterday regarding Alzheimer’s disease. As you may know, one of the big risk factors for Alzheimer’s is the gene for ApoE4, a particular subtype of the apolipoprotein gene (the healthier version is ApoE3). People with one copy of the ApoE4 gene have a single-digit multiple of the baseline, overall risk rate for the disease, and people with 2 copies have a many-fold (around 80) times increased risk.
It’s important to note that these are multiples of a “baseline risk” that is relatively small. This is a point often neglected when discussing the relative risks of a disease affected by particular risk factors when such information is conveyed to the general public. If the baseline risk for a disease were one in a billion (or less), then a four-times risk and an eighty-times risk might be roughly equivalent in the degree of concern they should raise. Eighty out of a billion is still less than a one in ten million chance for a disease; some other process would be much more likely to cause one’s deterioration and demise rather than the entity in question.
However, if the baseline risk were 1%‒a small but still real concern‒then a fourfold multiplier would increase the risk to one in 25. This is still fairly improbable, but certainly worth noting. An eighty-fold increase in risk would make the disease far more likely than not, and might well make it the single most important concern of the individual’s life.
Alzheimer’s risk in the general population lies between these two extremes, of course, and that baseline varies in different populations of people. Some of that variation itself may well be due to the varying frequency of the ApoE4 gene and related risk factors in the largely untested population, so it’s tricky to define these baselines, and it can even be misleading, giving rise to false security in some cases and inordinate fear in others. This is one example of how complex such diseases are from an epidemiological point of view, and highlight just how much we have yet to learn about Alzheimer’s specifically and the development and function of the nervous system in general.
Still, the article in question (I don’t have the link, I’m sorry to say) concerned one of the functions of the ApoE gene (or rather, its products) in general, which involve cholesterol transport in and around nerve cells. Cholesterol is a key component of cell membranes in animals, and this is particularly pertinent in this case because the myelin in nerves is formed from the sort of “wrapped up” membranes of a type of neural support cell*.
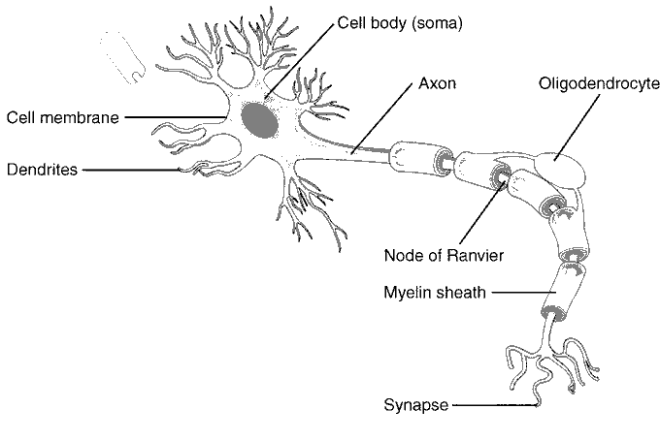
This particular study found that the cells of those with ApoE4 produced less or poorer myelin around nerve cells in the brain, presumably because of that faulty cholesterol transport, and that the myelin also deteriorated over time.
Now, the function of myelin is to allow the rapid progression of nerve impulses along relatively long axons, with impulses sort of jumping from one space (a “Node of Ranvier”) between myelin sheath and another rather than having to travel all the way down the nerve, which a much slower process, seen mostly in autonomic nerves in the periphery. When normally myelinated nerves lose their myelin, transmission of impulses is not merely slowed down, but becomes erratic and often effectively non-existent.

The researchers found that a particular pharmaceutical can correct for at least some of the faulty cholesterol transport and can thereby support better myelin survival. Though this does not necessarily point toward a cure or even a serious disease-altering treatment over the long term, it’s certainly interesting and encouraging.
But of course, we know Alzheimer’s to be a complex disease, and it may ultimately entail many processes. For instance, it’s unclear (to me at least) how this finding relates to the deposition of amyloid plaques, which are also related to ApoE, and are extracellular findings in Alzheimer’s. Are these plaques the degradation products of imperfect myelin, making them more a sign than a cause of dysfunction, or are they part of the process in and of themselves?
Also, it doesn’t address the question of neurofibrillary tangles, which are defects found within the nerve cells, and appear to be formed from aggregates of microtubule-associated proteins (called tau protein) that are atypically folded and in consequence tend to aggregate and not to function and to interfere with other cellular processes, making them somewhat similar to prions**. It’s not entirely clear (again, at least to me) which is primary, the plaques or the tangles, or if they are both a consequence of other underlying pathology, but they both seem to contribute to the dysfunction that is Alzheimer’s disease.
So, although potential for a treatment that improves cholesterol transport and supports the ongoing health of the myelin in the central nervous systems of those at risk for Alzheimer’s is certainly promising, it does not yet presage a possible cure (or a perfect prevention) for the disease. More research needs to be done, at all levels.
Of course, that research is being undertaken, in many places around the world. But there is little doubt that, if more resources were to be put into the study and research of such diseases, understanding and progress would proceed much more quickly.
The AIDS epidemic that started in the 1980s was a demonstration of the fact that, when society is strongly motivated to put resources into a problem, thus bringing many minds and much money to the work, progress can occur at an astonishing rate. The Apollo moon landings were another example of such rapid progress. Such cases of relative success can lead one to wonder just how much farther, how much faster, and how much better our understanding of the universe‒that which is outside us and that which is within us‒could advance if we were able to evoke the motivation that people have to put their resources into, for instance, the World Cup or fast food or celebrity gossip.
I suppose it’s a lot to expect from a large aggregate of upright, largely fur-less apes only one step away from hunting and gathering around sub-Saharan Africa that they collectively allocate resources into things that would, in short order, make life better and more satisfying for the vast majority of them. All creatures‒and indeed, all entities, down to the level of subatomic particles and up to the level of galaxies‒act in response to local forces. It’s hard to get humans to see beyond the momentary impulses that drive them, and this shouldn’t be surprising. But it is disheartening. That, however, is a subject for other blog posts.
I’ll try to have more to say about Alzheimer’s as I encounter more information. Just as an example, in closing, another article I found on the same day dealt with the inflammatory cells and mediators in the central nervous system, and how they can initially protect against and later worsen the problem. We should not be too surprised, I suppose, that a disease that leads to the insidious degeneration of the most complex system in the known universe‒the human brain‒should be complicated and multifactorial in its causation and in its expression. This should not discourage us too much, though. The most complicated puzzles are, all else being equal, the most satisfying ones to solve.
*The cell type that creates myelin in the peripheral nervous system (called Schwann cells) is different than the type that makes it in the central nervous system (oligodendrocytes), and this may be part of why Alzheimer’s affects the central nervous system mainly, whereas diseases like ALS (aka Lou Gehrig’s Disease), for instance, primarily affect the nervous system outside the brain.
**The overall shape of a protein in the body is a product of the ordering of its amino acids and how their side chains interact with the cellular environment‒how acidic or basic, how aqueous or fatty, how many of what ions, etc.‒and with other parts of the protein itself. Some proteins can fold in more than one possible way, and indeed this variability is crucial to the function of proteins as catalysts for highly specific chemical reactions in a cell. However, some proteins can fold into more than one, relatively stable form, one of which is nonfunctional. In some cases, these non-functional proteins interact with other proteins of their type (or others) to encourage other copies of the protein to likewise fold into the non-functional shape, and can form polymers of the protein, which can aggregate within the cell and resist breakdown, sometimes forming large conglomerations. These are the types of proteins that cause prion diseases such as “mad cow disease”, and they appear also to be the source of neurofibrillary tangles in people with Alzheimer’s disease.


Thank you for the lucid explanation of a subject I know very little about. I seem to recall that Bill Gates’s foundation is funding research into this disease (his father died of Alzheimer’s – if I can trust my memory).
You’re welcome. And I don’t know if that was deliberate wordplay there, but I liked it.